This is the first article of a four-part series on acid reflux and GERD. Read the second article on the myths of H. pylori and low stomach acid being the major causes, the third article on the mainstream medical treatments, and the final article on the myths of trigger foods and a GERD diet that works without drugs.
What are Acid Reflux and GERD?
About sixty million people in the US report that they suffer from painful symptoms of acid reflux at least once a week. Heartburn, abdominal pain, cough, regurgitation, sour taste in the mouth, sore throat, hoarseness, laryngitis, asthma-like symptoms, and sinus irritation are the typical symptoms. Smoking, pregnancy, obesity, hiatal hernia, and even tight-fitting clothes can make these symptoms worse.
Acid reflux occurs when acid and other contents of the stomach escape into the esophagus and beyond. Chronic acid reflux is often associated with damage because the esophagus and other affected tissues are not protected by the thick mucus layer that coats the inside of the stomach. And, the result is painful irritation which can lead to esophagitis, and Barrett’s esophagus, even esophageal cancer.
Persistent damaging reflux is referred to as gastroesophageal reflux disease (GERD). Acid reflux may also cause severe irritation of the vocal cords, a condition referred to as laryngopharyngeal reflux (LPR).
My Story
 For me this subject is personal. I am one of the sixty million people. I suffered from acid reflux for a good part of my adult life. I never understood what caused it. I only knew that my constant heartburn and regurgitation symptoms interfered with my work, my sleep, and my general wellbeing. My condition continued to get worse leading to anyone’s nightmare.
For me this subject is personal. I am one of the sixty million people. I suffered from acid reflux for a good part of my adult life. I never understood what caused it. I only knew that my constant heartburn and regurgitation symptoms interfered with my work, my sleep, and my general wellbeing. My condition continued to get worse leading to anyone’s nightmare.
I awoke in the middle of the night and leaped out of bed still half-dreaming, thinking “Oh my god, I’m dying. This is what it feels like to die.” I couldn’t breathe; I was choking for air and my lungs felt like they were filled with burning liquid. I ran to the bathroom coughing up something from my lungs that simply did not register. It felt like acid was filling my lungs. I soon realized that I had suffered severe acid reflux; stomach acid that traveled up my esophagus until I aspirated it into my lungs. I will never forget that moment. It defined the kind of fear and suffering that is possible with GERD.
Like many people with this condition, I recognized that my situation was becoming serious. But the recommended medicines – antacids, H2 blockers, and even PPI drugs did not give me adequate relief and I never noticed any dietary pattern that improved my symptoms. But that all changed in 2004.
 On the advice of my older son, a personal fitness trainer, I decided to go on a low-carb, high-fat diet (LCHFD) to lose a few pounds. What happened next made me forget about weight loss, because my reflux symptoms vanished the very next day. I couldn’t believe it! Why had no one ever told me about this? It turns out that most people didn’t know or were just finding out themselves. But thanks to Google, I soon realized that this was no fluke. Many others were saying the same thing and Drs. Mike and Mary Dan Eades had written about this in their bestselling book, Protein Power. I also found out that some doctors at Duke University had done a small clinical study confirming that cutting carbs can relieve symptoms.
On the advice of my older son, a personal fitness trainer, I decided to go on a low-carb, high-fat diet (LCHFD) to lose a few pounds. What happened next made me forget about weight loss, because my reflux symptoms vanished the very next day. I couldn’t believe it! Why had no one ever told me about this? It turns out that most people didn’t know or were just finding out themselves. But thanks to Google, I soon realized that this was no fluke. Many others were saying the same thing and Drs. Mike and Mary Dan Eades had written about this in their bestselling book, Protein Power. I also found out that some doctors at Duke University had done a small clinical study confirming that cutting carbs can relieve symptoms.
While the revelation was a great reward in and of itself, for the first time in my life I could control my symptoms, I was far from satisfied. I really wanted to know why. If removing most carbs stops heartburn, did that mean that carbs caused heartburn? I decided to get to the bottom of things and started doing research on how each type of food (fats, proteins, and carbohydrates) is digested. I read about and thought about the digestive process – chewing, salivary amylase for starch, stomach acid and pepsin for protein breakdown, lipase for fats, etc. And, as I started reading about digestion in the small intestine, a light bulb went off involving gut bacteria.
As a microbiologist, I had grown and studied a wide variety of bacteria for years in graduate school. I also worked with intestinal bacteria during a postdoc. at Tufts. While my focus was genetics, we constantly needed to grow enough bacteria to work with. And, I recalled two important traits of intestinal bacteria:
- They get the bulk of their energy from carbohydrates
- Most of them produce lots of gas
Depending on the strain, bacteria produce copious amounts of hydrogen and carbon dioxide gas. Also, intestinal Archaea, which look like bacteria under the microscope, but are actually a separate branch of life, produce a third intestinal gas, methane. Carbohydrates are the best fuel source for gas formation. 30 g of unabsorbed carbohydrates can allow bacteria to produce more than ten liters of hydrogen gas. [1] Imagine how much pressure this amount of gas would create in the intestines and this is only about one ounce of carbs. Intestinal bacteria produce so much gas that there have been well-documented cases of explosions during intestinal surgery.[2],[3] This gave me an idea:
“What if some of the carbs I had been eating were not being efficiently digested and absorbed into my blood stream, and instead they were persisting in my small intestine? Wouldn’t there be a chance that my acid reflux was the result of intestinal gas pressure from a bloom of gas-producing bacteria fed by carbohydrates? I.e. mentos in a coke bottle”
Could the answer be that simple? I needed to learn more and find out if this idea was full of holes. And, I set out to destroy my own theory : )
What we can agree on
The first thing I did was to learn all I could about what other scientists thought. One thing everyone seemed to agree on was that acid reflux occurs because the group of muscles at the top of the stomach are unable to keep the stomach’s contents from entering the esophagus. This group of muscles is called the lower esophageal sphincter (LES). The stomach’s contents include hydrochloric acid, bile and digestive enzymes. But why does acid reflux occur? According to the NIH:
“GERD results when the lower esophageal sphincter or LES becomes weak or relaxes when it should not, causing stomach contents to rise up into the esophagus.”
LES relaxation events are called transient lower esophageal sphincter relaxations (TLESRs). The LES appears to undergo these relaxation events, even when we are not swallowing. Both LES pressure and TLESRs can be measured by a technique called manometry, where a pressure-sensitive tube is passed down the esophagus through the LES. This technique has been used for many studies[4],[5],[6] and clearly shows that GERD patients exhibit:
- More episodes of reflux
- Less LES pressure
- More frequent episodes of TLESRs
- More extensive reflux during TLESRs, particularly after meals
- More pressure in the stomach (intragastric pressure) and more belching
I wondered if some of these ideas might disprove my theory or were they somehow consistent with my own observations. I thought about each observation to see if there was some common thread. Though increased pressure in the stomach had been well documented in GERD patients,[7] no one had a good explanation about where the pressure was coming from. But, my theory clearly explained a potential source: copious amounts of gas and pressure from bacterial fermentation in our intestines.
Got Gas?
 As for less LES pressure, you might be able to imagine intragrastric pressure “forcing” the LES to open as a possible mechanism of acid reflux. Clearly, if this were to occur, the LES pressure measurement would be reduced by the intragastric gas pressure. In other words, reduced LES pressure in GERD patients can be explained by my theory.
As for less LES pressure, you might be able to imagine intragrastric pressure “forcing” the LES to open as a possible mechanism of acid reflux. Clearly, if this were to occur, the LES pressure measurement would be reduced by the intragastric gas pressure. In other words, reduced LES pressure in GERD patients can be explained by my theory.
The same analogy can be applied to explain TLERs as well as increased reflux during TLESRs. The gas pressure forcing the LES to open would be measured as reduced LES pressure or relaxation events by manometry and be expected to increase the amount of refluxate in each event. This would be particularly true after meals if malabsorbed carbohydrates in each meal were driving the bacterial growth and gas production as I suspected. Thus, my theory also explains the increase in TLERs in GERD patients, particularly after meals.
While on the subject of gas pressure, there is one more important prediction my theory makes. If you were to stop the process of reflux and belching without resolving bacterial overgrowth, you might expect a buildup in gas pressure in the stomach and small intestine. One way to look at this question is to study people who have had fundoplication surgery, where the LES is surgically tightened, thus preventing reflux and belching. Reports on GERD patients who have undergone this procedure show that they suffer from new symptoms including excessive gas, flatulence, and bloating.[8],[9] The procedure is aimed at preventing reflux by tightening the LES muscles surgically, but the side effects are indicative of trapped stomach and intestinal gas as would be expected with uncontrolled malabsorption, and excessive bacterial fermentation.
Got bacteria?
 Essentially, I was proposing that excess dietary carbs could promote a kind of gut dysbiosis (unbalanced gut microbiota with too many gas-producing strains), possibly small intestinal bacterial overgrowth (SIBO). According to this idea, acid reflux occurs due to microbe-induced gas pressure. As I continued my research, I wondered what the world might look like if my theory was correct. The first thing I focused on was bacterial overgrowth itself. I reasoned that you should see a reduction in GERD symptoms whenever you reduced bacterial growth in the intestine.
Essentially, I was proposing that excess dietary carbs could promote a kind of gut dysbiosis (unbalanced gut microbiota with too many gas-producing strains), possibly small intestinal bacterial overgrowth (SIBO). According to this idea, acid reflux occurs due to microbe-induced gas pressure. As I continued my research, I wondered what the world might look like if my theory was correct. The first thing I focused on was bacterial overgrowth itself. I reasoned that you should see a reduction in GERD symptoms whenever you reduced bacterial growth in the intestine.
One way to control excessive bacterial growth in the intestine is to go on a low-carb diet because this type of diet limits carbohydrate fuel that bacteria need for growth. As I mentioned, low-carb diets reduce the symptoms of GERD as well as esophageal acid exposure.[10],[11] These results support my theory.
Similar to a low-carb diet, antibiotics can also reduce GERD symptoms, reduce esophageal acid exposure, and even prevent reflux into the lungs. [12],[13],[14] One plausible explanation for this effect is that the antibiotics help GERD by inhibiting the growth of gas-producing gut bacteria. Interestingly, antibiotics have been shown to increase LES pressure which is consistent with this idea.
My theory predicts that “overfeeding” gut microbes with hard-to-digest carbs in particular, will make GERD symptoms worse. Prebiotics represent a way to test this idea because prebiotics are carbohydrates that are not digested or absorbed. They are designed to feed the gut microbiota. But a study that looked at one prebiotic, fructose oligosaccharide (FOS), in GERD patients found that it caused more intestinal gas, increased GERD symptoms and increases in TLESRs.[15] A logical explanation is that the fermentation of FOS by gut bacteria creates excess gas pressure that drives (even more) reflux symptoms.
SIBO Connection
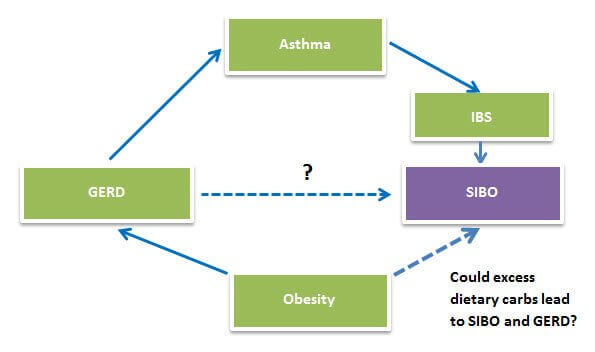
Lastly, I reasoned that if GERD involved SIBO, there would be an overlap in patient populations between GERD and other conditions known to involve SIBO including cystic fibrosis (CF), irritable bowel syndrome (IBS), obesity, and asthma.
Approximately 80% of people with cystic fibrosis (CF) also have GERD compared to about 15% percent in the general population.[16],[17],[18],[19] But that’s not all. CF patients also exhibit less LES pressure and more reflux episodes during TLESRs. Why is that? I think it has everything to do with SIBO.
People with CF exhibit well-documented carbohydrate malabsorption and SIBO.[20] The reason is that CF patients often have blockage of the pancreatic ducts by thick mucus, which blocks the release of the enzyme known as pancreatic amylase. Without this amylase, carbohydrate (starch) digestion and absorption do not occur in the small intestine. The lack of amylase results in more carbohydrates available for fermentation by gut bacteria, which leads to reflux.
 In GERD sufferers without CF, amylase release may not be blocked. But, for a variety of other reasons covered in the Fast Tract Digestion books (i.e. excess hard-to-digest dietary carbs, slowed motility, lactose intolerance, intestinal damage, etc.) I believe there also exists an oversupply of malabsorbed dietary carbohydrates. These excess carbs likely fuel excessive fermentation and induce reflux.
In GERD sufferers without CF, amylase release may not be blocked. But, for a variety of other reasons covered in the Fast Tract Digestion books (i.e. excess hard-to-digest dietary carbs, slowed motility, lactose intolerance, intestinal damage, etc.) I believe there also exists an oversupply of malabsorbed dietary carbohydrates. These excess carbs likely fuel excessive fermentation and induce reflux.
Approximately 50% of the people who have IBS also suffer from GERD and almost as many GERD patients suffer from IBS. This percentage is about three times higher than what is seen in the general population. IBS has been clearly linked to SIBO, and like GERD, has been treated successfully with carbohydrate restriction as well as antibiotics.[21],[22],[23],[24],[25] This evidence is consistent with SIBO playing a role in both conditions.
GERD, Obesity, and Asthma:
Obesity is tightly linked to GERD. Obese individuals generally consume more food, especially carbohydrates. You might predict that the increase in dietary carbohydrates would increase malabsorption leading to SIBO and reflux. Interestingly, carbohydrate restriction improves GERD symptoms and reduces esophageal acid exposure in obese patients whether or not they lose weight![26]
Asthma is connected to SIBO by way of IBS. A study of people with asthma showed that they had IBS twice as often as non-asthmatics.[27] And, asthma is even more tightly linked to GERD. As many as 80% of asthmatics suffer from abnormal gastroesophageal reflux compared to about 20% of non-asthmatics.[28], [29] Several large studies showed that acid-reducing drugs don’t improve asthma symptoms, but stopping reflux itself with fundoplication operations dramatically improves asthma symptoms. And, this results in a significant reduction in medicine usage.[30],[31] This suggests to me that something in reflux other than acid exacerbates asthma. Perhaps bile, bacteria and/or enzymes could be responsible. Leaky gut, another condition linked to SIBO, may be a factor in asthma as well, but the fundoplication data leads me to favor acid reflux as the most significant trigger in the exacerbation of asthma. It could even be an etiological cause of asthma. For more info, check out my blog on the Heartburn-Asthma link.
More recently, SIBO has actually been detected in GERD patients. Half of the GERD patients showed evidence of SIBO by glucose breath testing.[32] Eighty-seven to ninety percent of SIBO-positive patients showed improvement after antibiotic treatment. The study design makes it difficult to draw final conclusions regarding SIBO and GERD however. The study included IBS patients and everyone in the study was taking PPI drugs, known to exacerbate SIBO. Also, the less sensitive “glucose” breath test method was used.
I believe the number of SIBO-positive results in both IBS and GERD patients would have been higher if the study employed the more sensitive lactulose breath test instead of the glucose. Because lactulose is not digested or absorbed in the small intestine, it can detect bacteria throughout the entire length of the small intestine. Glucose on the other hand, is rapidly absorbed in the first part of the small intestine and will only detect bacteria if they are present in this region.
Summary
Instead of “destroying my theory”, all the evidence I looked at supported my idea: carbohydrate malabsorption and bacterial overgrowth is the root cause of acid reflux. But, how about other proposed causes? For instance, H. pylori and low stomach acid? My research has led me to some interesting findings. I examined both in my second article, “Is GERD caused by H. pylori & Low Stomach Acid?”.
Read the third article, “GERD – Why standard treatments are ineffective”.
Read the final article, “GERD diet that works without drugs”.
______________________________________________________________
[1] Suarez F, Levitt M. Textbook of Primary and Acute Care Medicine. Second edition. Edited by Gideon Bosker. Page 1191. [2] Dener IA, Demirci C. Explosion during diathermy gastrotomy in a patient with carcinoma of the antrum. Int J Clin Pract. 2003 Oct; 57(8):737-8. [3] Bigard M-A, Gaucher P, Lassalle C. Fatal colonic explosion during colonoscopic polypectomy. Gastroenterology 1979; 77: 1307-1310. [4] Iwakiri K, Hayashi Y, Kotoyori M, Tanaka Y, Kawakami A, Sakamoto C, Holloway RH. Transient lower esophageal sphincter relaxations (TLESRs) are the major mechanism of gastroesophageal reflux but are not the cause of reflux disease. Dig Dis Sci. 2005 Jun;50(6):1072-7. [5] Dodds WJ, Dent J, Hogan WJ, Helm JF, Hauser R, Patel GK, Egide MS. Mechanisms of gastroesophageal reflux in patients with reflux esophagitis. N Engl J Med. 1982 Dec 16;307(25):1547-52. [6] Trudgill NJ, Riley SA. Transient lower esophageal sphincter relaxations are no more frequent in patients with gastroesophageal reflux disease than in asymptomatic volunteers. Am J Gastroenterol. 2001 Sep;96(9):2569-74. [7] Dodds WJ, Dent J, Hogan WJ, Helm JF, Hauser R, Patel GK, Egide MS. Mechanisms of gastroesophageal reflux in patients with reflux esophagitis. N Engl J Med. 1982 Dec 16;307(25):1547-52. [8] Vakil N, Shaw M, Kirby R. Clinical effectiveness of laparoscopic fundoplication in a US community. Am J Med. 2003 Jan;114(1):1-5. Klaus A, Hinder RA, DeVault KR, Achem SR. Bowel dysfunction after laparoscopic anti reflux surgery: incidence, severity, and clinical course. Am J Med. 2003 Jan;114(1):6-9. [9] Beldi G, Gláttli A. Long-term gastrointestinal symptoms after laparoscopic Nissen fundoplication. Surg Laparosc Endosc Percutan Tech. 2002 Oct;12(5):316-9. [10] Yancy WS Jr, Provenzale D, Westman EC. Improvement of gastroesophageal reflux disease after initiation of a low-carbohydrate diet: five brief cased reports. Altern Ther health med. 2001. Nov-Dec; 7(6):120,116-119. [11] Austin GL, Thiny MT, Westman EC, Yancy WS Jr, Shaheen NJ. A very low-carbohydrate diet improves gastroesophageal reflux and its symptoms. Dig Dis Sci. 2006 Aug;51(8):1307-12. [12] Pennathur A, Tran A, Cioppi M, Fayad J, Sieren GL, Little AG. Erythromycin strengthens the defective lower esophageal sphincter in patients with gastroesophageal reflux disease. Am J Surg. 1994 Jan;167(1):169-173. [13] Pehl C, Pfeiffer A, Wendl B, Stellwag B, Kaess H. Effect of erythromycin on postprandial gastroesophageal reflux in reflux esophagitis. Dis Esophagus. 1997 Jan;10(1):34-37. [14] Mertens V, Blondeau K, Pauwels A, Farre R, Vanaudenaerde B, Vos R, Verleden G, Van Raemdonck DE, Dupont LJ, Sifrim D. Azithromycin reduces gastroesophageal reflux and aspiration in lung transplant recipients. Dig Dis Sci. 2009 May;54(5):972-9. [15] Piche T, des Varannes SB, Sacher-Huvelin S, Holst JJ, Cuber JC, Galmiche JP. Colonic fermentation influences lower esophageal sphincter function in gastroesophageal reflux disease. Gastroenterology. 2003 Apr;124(4):894-902. [16] Ledson MJ, Tran J, Walshaw MJ. Prevalence and mechanisms of gastro-oesophageal reflux in adult cystic fibrosis patients. J R Soc Med. 1998 Jan;91(1):7-9. [17] Vic P, Tassin E, Turck D, Gottrand F, Launay V, Farriaux JP. Frequency of gastroesophageal reflux in infants and in young children with cystic fibrosis. Arch Pediatr. 1995 Aug;2(8):742-6. [18] Fridge JL, Conrad C, Gerson L, Castillo RO, Cox K. Risk factors for small bowel bacterial overgrowth in cystic fibrosis. J Pediatr Gastroenterol Nutr. 2007 Feb;44(2):212-8. [19] Pauwels A, Blondeau K, Dupont LJ, Sifrim D. Mechanisms of increased gastroesophageal reflux in patients with cystic fibrosis. Am J Gastroenterol. 2012 Sep;107(9):1346-53. [20] Lisowska A, Wójtowicz J, Walkowiak J. Small intestine bacterial overgrowth is frequent in cystic fibrosis: combined hydrogen and methane measurements are required for its detection. Acta Biochim Pol. 2009;56(4):631-4. [21] Pimentel M, Chow EJ, Lin HC. Eradication of small intestinal bacterial overgrowth reduces symptoms of irritable bowel syndrome. Am J Gastroenterol. 2000;95:3503-6. [22] Austin GL, Dalton CB, Hu Y, Morris CB, Hankins J, Weinland SR, Westman EC, Yancy WS Jr, Drossman DA. A very low-carbohydrate diet improves symptoms and quality of life in diarrhea-predominant irritable bowel syndrome. Clin Gastroenterol Hepatol. 2009 Jun;7(6):706-708. [23] Majewski M, Reddymasu SC, Sostarich S, Foran P, McCallum RW. Efficacy of rifaximin, a non absorbed oral antibiotic, in the treatment of small intestinal bacterial overgrowth. Am J Med Sci. 2007 May;333(5):266-70. [24] Pimentel M. Review of rifaximin as treatment for SIBO and IBS. Expert Opin Investig Drugs. 2009 Mar;18(3):349-58. [25] Yang J, Lee HR, Low K, Chatterjee S, Pimentel M. Rifaximin versus other antibiotics in the primary treatment and retreatment of bacterial overgrowth in IBS. Dig Dis Sci. 2008 Jan;53(1):169-74. [26]Hagen J, Deitel M, Khanna RK, Ilves R. Gastroesophageal reflux in the massively obese. Int. Surg. 1987 Jan-Mar;72(1):1-3. Fisher BL, Pennathur A, Mutnick JL, Little AG. Obesity correlates with gastroesophageal reflux. Dig Dis Sci. 1999 Nov;44(11):2290-4. Austin GL, Thiny MT, Westman EC, Yancy WS Jr, Shaheen NJ. A very low-carbohydrate diet improves gastroesophageal reflux and its symptoms (Obese patients). Dig Dis Sci. 2006 Aug;51(8):1307-12. [27]Roussos A, Koursarakos P, Patsopoulos D, Gerogianni I, Philippou N. Increased prevalence of irritable bowel syndrome in patients with bronchial asthma. Respir Med. 2003 Jan;97(1):75-9. [28]Sontag SJ, O’Connell S, Khandelwal S, Miller T, Nemchausky B, Schnell TG, Serlovsky R. Most asthmatics have gastroesophageal reflux with or without bronchodilator therapy. Gastroenterology. 1990 Sep;99(3):613-20. [29]Leggett JJ, Johnston BT, Mills M, Gamble J, Heaney LG. Prevalence of gastroesophageal reflux in difficult asthma: relationship to asthma outcome. Chest. 2005 Apr;127(4):1227-31. [30] Rothenberg S1, Cowles R. The effects of laparoscopic Nissen fundoplication on patients with severe gastroesophageal reflux disease and steroid-dependent asthma. J Pediatr Surg. 2012 Jun;47(6):1101-4. [31] Spivak H1, Smith CD, Phichith A, Galloway K, Waring JP, Hunter JG. Asthma and gastroesophageal reflux: fundoplication decreases need for systemic corticosteroids. J Gastrointest Surg. 1999 Sep-Oct;3(5):477-82. [32]Lombardo L, Foti M, Ruggia O, Chiecchio A. Increased incidence of small intestinal bacterial overgrowth during proton pump inhibitor therapy. Clin Gastroenterol Hepatol. 2010 Jun;8(6):504-8







Have you ever investigated the link between GERD and histamine intolerance?
Hi NoGlutenEver,
No, I have not. I do recognize that histamine released from gastric mucosal cells (ECL cells) stimulates the acid-making cells (parietal cells) to release acid and that H2 blockers stop acid release by blocking their response to histamine. So it seems possible that high histamine levels might result in more acid release. Do you have some other ideas on histamine intolerance?
Could explain some seasonal variation in GERD symptoms. Could also be part of the reason why some people do not fare will with a Paleo type diet especially if they use cook ahead techniques to consolidate food prep, such as in the Well Fed cookbooks.
Winter foods, especially holiday treats, can be high in histamine. I am thinking about smoked meats and fish, cheeses, things that you wouldn’t necessarily suspect in GERD issues. Pumpkin pie too. Also foods that are slow cooked, or deliberately served the next day (chili, roasts) for better flavor.
Apparently seasonal allergies can “use up” your histamine tolerance “bucket.” And if you take anti-histamines for allergy relief, they only block 2 of the 4 types of histamine receptors. The histamine is still there so it can overload the remaining receptors which cause a different reaction.
Another seasonal variation might be exercising in extreme weather, which would use up some of the threshold also.
Many people with histamine issues see it because it shows up as skin issues, but heartburn is also a symptom apparently. Also headache, anxiety. The recent Kresser article got me to thinking about this. https://chriskresser.com/headaches-hives-and-heartburn-could-histamine-be-the-cause
Apparently some foods can help inhibit histamine release, so looking a X and Y foods may be less helpful than looking at the whole menu.
Definitely a YMMV sort of topic, but I thought you might be interested. If some one is seeing limited or inconsistent improvement with the Fast Track approach, it may be worth a look.
Interesting idea. I took a look at Chris’ and Joneja’s articles and the references for the latter but could not find any primary data linking acid reflux to histamine levels. Did I miss it?
I’ve not gotten much beyond this:
https://www.ncbi.nlm.nih.gov/pubmed/11246041
and this has some links also
https://www.vivo.colostate.edu/hbooks/pathphys/digestion/stomach/parietal.html
cortisol and histamine:
https://onlinelibrary.wiley.com/doi/10.1111/j.1476-5381.1972.tb08102.x/abstract
more on stress (psych aspects)
https://www.medscape.com/viewarticle/588970_3
Are histamine reactions inconsistent?
https://www.ncbi.nlm.nih.gov/pubmed/21165702
and what’s the half life, anyway?
https://www.spirxpert.com/brrespuk/duration.htm
https://link.springer.com/article/10.1007%2FBF01980899#page-1
This is getting above my pay grade, that’s for sure. I found this overview to be helpful and it does link to studies:
https://healthypixels.com/?p=1044
and an interesting tidbit –
What do GERD and ERD (Erectile Dysfunction, not Erosive Reflux) have in common? the H2 receptor.
Thanks for your blog and books and interaction with your readers; very helpful, life changing, really.
Wow, thanks for the links. I will take a look at these when I have a moment.
Your system seems to be helping me with LPR and night time GERD. I am also finding chewable DGL from Source Naturals to be helpful before meals. I struggle with constipation but have found Calm magnesium citrate is helping with that. I am a little concerned about the lack of fiber for long term lower digestive health though. Chris Kresser, whom mentioned you in one of his articles, isn’t pro high fiber but did say very low fiber from low carb diets may be a problem long term. I respect both of you very much. Thoughts?
Happy to hear about your progress with LPR and night time GERD Jill. I understand that many people believe that consuming lots of fiber is healthy and not getting enough can give you every health problem under the sun including cancer. I used to believe that myself. But as I found out, and wrote about in my fiber blog series, the health claims are not supported and the dark side of fiber for digestive health is real. As for constipation, you might be interested in this recent paper reporting that “no fiber” is far superior to “moderate or high fiber” for constipation.
When I developed the Fermentation Potential formula and started calculating FP values for various foods, I realized that there is much more fermentable material in our diet than was previously realized. And the FP calculation doesn’t include animal sources which is now known to further contribute to this fermentable material available to our gut microbes. I believe that 30-40 grams of FP per day is plenty to keep out microbes happy but not enough to make those of us with low tolerance ill.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3435786/
Great paper you linked on ‘no fiber’, and full text, too!
Hope you are sitting down, because I am going to finally agree that no or low fiber is a great intervention for many people. There are just so many combinations of problems that cause people gastric distress that you can’t discount using low/no fiber.
The paper chronicals 62 people battling chronic constipation. The ones who adhered to a no-fiber diet were cured.
I would love to see a breakdown of their diet and if it was truly ‘no fiber.’ I really can’t imagine a ‘no fiber’ diet…steak and eggs? Ketogenic? Certainly contained zero wheat or grains and most fruit/veggies. When I was LC/VLC I was probably eating 5-10g in fibers, and at that point, my stools were consistently Bristol Scale 1 (rabbit poop!).
I think for healthy people with no digestive issues to speak of, 20-40g of fiber with the bulk from RS or inulin is very healthy. But if there is significant dysbiosis, and fiber restriction helps, that is clearly a superior intervention compared to meds or surgery.
Hey Tim,
Thank god I was setting down. I need oxygen. Can I get some oxygen here?
So we agree that limiting fermentable material (if you accept fiber, why not other hard to digest carbs which behave like fiber) can help people avoid gastric distress.
What I disagree with is the characterization of people who don’t tolerate excessive fiber and other fermentable carbs as being “unhealthy”. If someone goes through life completely healthy and happy simply by matching their fermentable carb intake to their tolerance level, I don’t see them as ill, sick or unhealthy. Perhaps some people simply evolved to eat a diet lush with animal foods and almost every vegetable (most are very low in fermentable carbs) as well as some seasonal fruit, but simply less fiber, resistant starch, fructose and lactose.
“If someone goes through life completely healthy and happy simply by matching their fermentable carb intake to their tolerance level, I don’t seem them as ill, sick or unhealthy.”
That pretty much sums it up! People owe it to themselves to keep trying until they find the right combination that keeps them at a healthy weight, pooping good, and not in pain.
Great to have reached this understanding Tim.
Correction on the supplements listed in my previous post. I take Natural Factors DGL and Natural Calm. Sorry for any mix up for anyone.
Hi Norm
I have your fast tract book, and have been following the diet for approx 2 months. I have LPR, and have found that a very parred down (simplified) version of the diet is leading to greatly reduced symptoms. Whilst I do still have symptoms each day, to varying degrees, I am now off PPIs to my great delight. For the first month of the diet i was on heavy duty anti biotics (post operative). This may be complicating things, i just dont know.
I have two questions. The first relates to being tested for SIBO. I have finally(!!!!!!!!!) found a dr who will refer me to be tested for sibo. This test will take place in two weeks. The delay is down to the fact i have been taking probiotics, and need to abstain from these for a two week period before taking the test. My question is, will the reduced sibo as a result oc the diet influence the test results? Should i eat some resistent starches prior to the test ? My secong question is one i know you have answered, but i cant find your response in the threads. My question is what ingredients should i avoid in sugar free gum? I am in australia, so brand names differ from USA (also makes some recipes difficult sadly).
I submitted the above post without finishing… I would like to thank you Norm, and all the other people who post on this site. You have helped me in so many ways, not least by inspiring me to get of PPIs. Thank goodness!!!!
I appreciate your post Pamela. Great news that you have been able to get off PPIs! The Fast Tract Diet will very likely suppress any SIBO you may have had. But why feed it with resistant starch just to make the test positive.
Yes, we did have some questions about chewing gum. I recall that a stick has an FP of 1 gram. Someone mentioned a chewing gum (Australian I believe) made with Splenda. I did a search and could not find it in the states.
My rational iS wanting a definitive yes or no as to whether SIBO has been the issue. If it is i will know i am on the right track, but need more time. The diet is working to a degree, but i definately continue to have symptoms. I have also had to greatly simplify the diet.
I see. That’s a “Catch 22”.
Hi Norm
I think I need to have a conversation with you. Do you do consults with pople in Australia?
Yes, we use Skype. I will have someone contact you Pamela.
Dear Dr Robillard,
I suffer from terrible GERD and have recently bought and read Fast Tract Digestion Heartburn, it makes a lot of sense and I hope it will help keep my symptoms under control. I was hoping you would be able to answer a couple of questions I have. Firstly, I am really confused about whether taking probiotics when you have SIBO is a good idea or not. On the one hand you have doctors like Dr Pimentel who say that they should be avoided as they aggravate the situation, and on the other hand doctors like Dr Siebecker seem to think that they can be helpful in most cases. It’s hard to know who to believe! I couldn’t find a clear and definitive answer in your book, what is your opinion on the matter? Secondly, how do you feel about the theory that GERD sufferers should avoid fatty foods as they cause delayed stomach emptying, thereby exacerbating reflux? Many thanks for your help!
Hi Nathalie,
For GERD, the safest probiotic are strains, such as Lactobacillus acidophillus and Bifidobacteria, that don’t make gas from carbs. Many of the other strains do make gas from carbs. Until more advanced probiotics are available, I would stick to products just containing these two strains for GERD.
I believe fats to be the safest choice for GERD, with carbs being the least safe and proteins somewhere in the middle. That’s because fats are least fermentable resulting in less gas production. It’s all about the gas.
Hi Nathalie,
There are several things to consider with probiotics for SIBO and each person’s unique situation must be evaluated. We cover this in our consultation program. As for your question on fats. This question is covered (it’s the first entry) on the Fast Tract Digestion Q&A page you can find here. https://digestivehealthinstitute.org/fast-tract-diet-qa/
Hi Norm,
I hope you are doing well. I have a quick question please about Beta Glucans. I just realized that these are in the Immune supplements I take by Researched Nutritionals. I have Lyme Disease and was diagnosed a year ago today. I am doing better so I was wondering what you thought about the types of Beta Glucans that are in these products and how they might affect my gut. I am doing a little better, too, but my stomach is still poofed out :( I am a thin person so I think it looks bigger because of that.
https://www.researchednutritionals.com/store/item.cfm?code=CRN111
https://www.researchednutritionals.com/store/item.cfm?code=CRN116&cat=3
https://www.researchednutritionals.com/store/item.cfm?code=CRN114&cat=3
those are three I take.
I see this one just has the transfer factors without the other stuff so maybe my doc. might allow me to take this one instead-
https://www.researchednutritionals.com/store/item.cfm?code=CRN123
Your feedback would be appreciated since this is your specialty
I know it gets more complex when you have multiple issues going on that aren’t all caused by the gut
Thanks :)
take care
Hi Lori,
Beta glucan is a dietary fiber, but looks like there is no more than about 1/2 a gram. I wouldn’t worry about it.
Thank you so much.
Take Good Care.
Lori
It all makes so much sense now! Thank you for your article. I’m still nervous at how to proceed at this point, but I’m looking at getting your heartburn book. Does the book explain how a person can wean off the PPI’s and get to the point where the GERD is controlled by diet alone? Is the diet something that has to be followed forever or does it start out super strict and then get a tad bit easier to follow? I had previously given up on the diet route because I have 2 little ones in diapers and not much time on my hands, but now the doctors seem to have no more answers and things only get worse. My acid is basically controlled with the PPI’s, but I still get reflux that wakes me up coughing or just with trouble breathing. The more sugar, starches and grains I eat, the worse it is. I keep telling the GI doc that, but they don’t seem to get the correlation. They just seem to think I have a faulty LES and keep telling me that my problem is that my LES is relaxing sporadically. Will your heartburn book help me and is this a diet that is possible for a mom of 2 whose husband is working day and night and has very little free time? Are there recipes that I can access if I get this book and is there an extensive food list or at least a very comprehensive way of learning how to determine what foods are ok? I know I will be cutting sugar altogether, but does that include the natural sugar occurring in fruit or honey? Is dairy to be avoided? Thanks so much for your time! Just trying to see if this book will be both doable and worth it for me. I’m currently looking at this book, the SCD diet and the GAPS diet and trying to determine what will be best. Thanks!
Hi Andrea,
The answers, lists, recipes and discussions on PPIs are all in book. I think it would be a worthwhile read given your situation.
Sorry, I was still wondering more about the diet. As I asked in my comment above, is this diet something that is fesable for a very busy mom to follow? Will it make me loose weight? (that would be a bad thing for me) Is this diet even more strict than the scd diet? And lastly, can you direct me to the research that shows this is better than scd or gaps or any of the other diets out there that claim to heal gerd? I just have to know these things before spending more money trying to fix my gerd. I just want to be sure this book is the right one when there are so many others out there. Is there somewhere I can learn more about the diet and what is excluded? I just need to know if this is something possible for me to do before I go out and get the book. Thanks!
Dr. Robillard,
Fascinating research and theory – based on personal experience I’m convinced that your “bacterial culprit” proposition for a range of digestive issues is spot on.
I have had numerous digestive problems all my life, and after 40 years I also found out that I have Celiac disease.
I recovered well on the gluten free diet, and symptoms gradually disappeared.
Recently though – after a highly irresponsible binge-period involving significant amounts of candy, soda, coffee, sweets and stress (I don’t know what was I thinking) – I developed pretty nasty LPR, one new symptom I never had before.
I came across your research and books, and they immediately “clicked”.
I started the diet (no strict portioning, just watching ingredients / FP so far), and it lead to remarkable improvement in as few as 4-5 days.
However, right now I’m struggling with a relapse, which I have somewhat of a hard time placing and interpreting.
I suspected a number of things:
– watermelon (which I absolutely love, and would be devastated to lose)
– a bit of taco sauce, which I discovered contained HFCS
– some corn-based gluten free pasta
– possibly too large portions…
…but I don’t have a clear “winner” yet.
A gluten free diet can be challenging enough, and combined with low FP it’s been tricky to not feel perpetually hungry. :)
All in all, I’m wondering what your experience has been with the duration of occasional “setbacks”?
Thank you in advance for any insight.
Hi Ed,
I would first look at the gluten-free pasta – too much resistant starch. After that, consider fructose intolerance and reduce those foods, even cutting back on the watermelon. One other trick you might try if you want to keep some fructose-containing foods, is adding a bit of powdered dextrose (glucose) when you have fructose. Glucose improves absorption of fructose. Good luck!
How does this study tie in with a dietary model for control/cure of GERD?
de Souza Pereira R. “Regression of gastroesophageal reflux disease symptoms using dietary supplementation with melatonin, vitamins and amino acids: comparison with omeprazole.” J.Pineal Res. 2006; 41:195–200
I would like to see this work reproduced by another lab.
I think you make a very important comment ; “What if some of the carbs I had been eating were not being efficiently digested and absorbed into my bloodstream, and instead they were persisting in my small intestine ?”
One has to consider and focus on the fact that the average American has lost 40% of the diversity of their human microbiome.
This loss of species is being connected with a long list of diseases. We very likely have lost important species in the small intestine, and this may be connected with your hypothesis. I suspect it is.
How do we go about restoring these missing species ? First and foremost, we have to stop the continuing damage. I’ll list some of the culprits : mouthwash (kills bacteria), toothpaste (a low dose poison to bacteria), chlorine (kills bacteria), Roundup containing foods(kills our good bacteria), emulsifiers in processed foods (cause inflammation), antibacterial hand soaps and antibacterials in other common products like scissors, etc. , chemicals in antiperspirants, soaps, dryer sheets, and more, pesticide and herbicide residues, over the counter supplements, and medicines. Eliminating as many of these damaging things is necessary before we can even start to restore diversity.
The correct diet ; diets high in fresh fruits and fresh vegetables are good for our good, helpful microbes. We should supplement these diets with good probiotic fermented foods such as Kefir, Kimchi, Kombachu, Sauerkraut, and Miso, etc.. These products have good, beneficial microbes, the ones we want. Cooked vegetables are OK. Avoid all processed foods, as many have emulsifiers which cause inflammation to the microbiomie. Avoid all sugars, as sugars feed our bad bacteria. Avoid all products that may have Roundup in them, as Roundup is killing our good, beneficial microbes and is causing inflammation.
Good points on diversity Joe. I don’t have problem with most of your recommendations, but personally I have found that reducing, not increasing fermentable carbs including prebiotics is the best approach for my symptoms and supported by the ideas and references in this article.
I see no mention of Hiatal Hernia in your latest book I just purchased.
If you have a hiatal hernia, no amount of diet is going to assist with the low LES pressure caused by a mechanical problem. Do you have any thoughts on this?
Also, I had GERD 10 years ago I somehow got rid of, but has now been back with 24/7 heartburn for over 2 months with seemingly nothing helping. PPI does not seem to help with the heartburn which indicates possible bile reflux and issues with liver/gall bladder, also something not mentioned, any thoughts? I am rather desperate.
I have read Stress can cause SIBO itself or low stomach acid, which in turn causes SIBO and then possibly GERD
Good call Nick. I should add something on this to the next edition. I see two issues with HH. One involved some additional potential extragastric pressure (like you might imagine with pregnancy or a tight belt) since part of the stomach is getting pinched. This translated in to more intragastric pressure. The Fast Tract Diet should help since the approach focuses on reliving intragastric pressure. But there are reports that HH may actually affect the operation of the LES directly. While less intragastric pressure is still helpful, there may be situations were correction of the HH is appropriate. Your thoughts?
Hi Dr. Robillard,
I have had heartburn now for a couple of years, and am very eager to try out the low-carb method. It was interesting reading your theory, which makes much more sense than other medical advice I’ve seen online. Out of curiosity, I wonder if you would have any ideas about how acid reflux could relate to the female hormonal cycle? I have consistently experienced that my worst problems with heartburn coincide with PMS. Would this be psychological/stress related, or does hormones affect the growth of bacteria?
Interesting connection Christa. I’m afraid I don’t have an answer on this one. Please share anything you are able to find out.
Hi Christa, your comment on hormones is interesting and I can suggest something worth looking into is chemical sensitivities (naturally occurring in foods and as a result of fermentation) as these do increase with hormone levels. This is coming from migraine information rather than IBS or GERD. Migraine can also affect your stomach (it’s even possible to have a stomach migraine and no headache) so do have a look into it and see if you find an answer that applies to you.
Hi again Christa and Norm – I found this on hormones you might find interesting: “Like many other things, production of DAO fluctuates with the menstrual cycle: it’s higher during the luteal phase (starting roughly a week after your period ends, and lasting until the start of your next period) than during the follicular phase (the rest of the time). This means that histamine intolerance issues will likely be worse during the follicular phase (while you’re bleeding and then for a week afterwards), and show some improvement at other times, regardless of what you do or don’t eat. With this monthly fluctuation, it’s even harder to figure out what foods you might be reacting to.”
Hi Norm. This morning is the first time I have come across your research. Last night I was reading all about laryngopharyngeal reflux (LPR) as it explains exactly what’s been happening to me the last few months. Someone who suffers from LPR commented on a blog and said she had great success with the Fast Track Diet so I looked it up and here I am now. I have had digestive issues for as long as I can remember and especially since 19 years old. About ten years or more ago I was diagnosed with GERD but PPIs and H2 blockers just made me feel nauseous after the initial few days relief so I discontinued them. I also didn’t believe blocking acid was the answer. Eventually I discovered the Royal Prince Alfred Hospital’s research and easy to use booklet on food sensitivities or intolerances (ie Amines and Salycilates and MSG) and have more or less followed that for a very long time as it’s been the most helpful approach. I found it because a Naturopath in a health food store gave me his best guess allergy elimination/safe foods list which I followed for a couple of weeks and felt a lot better than I had for a long time. My gut pain (chronic non ulcer dyspepsia my GP called it) symptoms eased off so I researched further. At that time there was nowhere near all the confusing and conflicting approaches there are now. Recently I noticed foods that are both High FodMap and High in Histamines seem to be triggers for me. In the past few days I decided to cut out all sweet biscuits, cakes and chocolate (chocolate I have avoided for many years in the past). I wondered why the FodMap researchers were not also considering chemical sensitivities. On this thread I can see that someone has drawn Histamines to your attention. They are definitely culprits for me, along with MSG (biggest triggers are brown meat gravy, slow cooked red meat and left over Chinese food, but I also avoid or limit many other foods as too much of a tasty thing usually ends in pain and all kinds of unwelcome symptoms). I can’t ignore food sensitivities as I also suffer from migraine so I will have to find ways to combine all these different approaches!